
ТОР 5 статей:
Методические подходы к анализу финансового состояния предприятия
Проблема периодизации русской литературы ХХ века. Краткая характеристика второй половины ХХ века
Характеристика шлифовальных кругов и ее маркировка
Служебные части речи. Предлог. Союз. Частицы
КАТЕГОРИИ:
- Археология
- Архитектура
- Астрономия
- Аудит
- Биология
- Ботаника
- Бухгалтерский учёт
- Войное дело
- Генетика
- География
- Геология
- Дизайн
- Искусство
- История
- Кино
- Кулинария
- Культура
- Литература
- Математика
- Медицина
- Металлургия
- Мифология
- Музыка
- Психология
- Религия
- Спорт
- Строительство
- Техника
- Транспорт
- Туризм
- Усадьба
- Физика
- Фотография
- Химия
- Экология
- Электричество
- Электроника
- Энергетика
Задачи для самостоятельного решения 3 страница
** Продукты сгорания: CO2, Cl2 (г), HCl (р-р).
*** Продукты сгорания: CO2, H2O (ж), HCl (р-р).
Приложение 12
Энтальпии образования веществ, стандартные
энтропии, температурные коэффициенты в уравнении
теплоемкости и стандартные изобарные теплоемкости
веществ
| Вещество |  кДж/моль кДж/моль
| 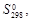 Дж/(моль·К)
Дж/(моль·К)
| Коэффициенты уравнения 
| 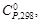 Дж/(моль·К)
Дж/(моль·К)
| ||||||
| а | b ×103 | с' ×10–5 | ||||||||
| Простые вещества и неорганические соединения Формула для вычисления теплоемкости Ср 0 = а + bТ + с1Т -2 | ||||||||||
| Ag (т) | 42,55 | 23,97 | 5,27 | –0,25 | 25,44 | |||||
| AgNO3-a (т) | –124,52 | 140,92 | 36,65 | 189,12 | – | 93,05 | ||||
| Ag2O (т) | –30,54 | 121,75 | 55,48 | 29,46 | – | 65,86 | ||||
| Ag2S-a(т) | –31,80 | 143,51 | 64,60 | 39,96 | – | 76,53 | ||||
| Ag2SO4(т) | –715,88 | 200,00 | 96,65 | 116,73 | – | 131,38 | ||||
| AgBr(т) | –100,42 | 107,11 | 33,18 | 64,43 | – | 52,30 | ||||
| AgCl(т) | –126,78 | 96,23 | 62,26 | 4,18 | –11,30 | 50,79 | ||||
| AgI-a(т) | –61,92 | 115,48 | 24,35 | 100,83 | – | 54,43 | ||||
| Ag3PO4(т) | –990,0 | 258,2 | – | – | – | – | ||||
| Ag2CO3(т) | –506,0 | 167,0 | – | – | – | – | ||||
| Al (т) | 28,33 | 20,67 | 12,39 | – | 24,35 | |||||
| Al2O3 (корунд) | –1675,0 | 50,94 | 114,56 | 12,89 | –34,31 | 79,04 | ||||
| Al2(SO4)3(т) | –3441,80 | 239,20 | 366,31 | 62,60 | –112,5 | 259,41 | ||||
| AlF3(т) | –1510,42 | 66,48 | 72,26 | 45,86 | –9,62 | 75,10 | ||||
| AlCl3(т) | –704,17 | 109,29 | 77,12 | 47,83 | – | 91,00 | ||||
| AlBr3(т) | –513,38 | 180,25 | 49,95 | 169,58 | – | 100,50 | ||||
| AlI3(т) | –308,0 | 189,5 | – | – | – | – | ||||
| Al(OH)3(т) | –1294,0 | 70,1 | – | – | – | – | ||||
| As(т) | 35,61 | 23,28 | 5,74 | – | 24,74 | |||||
| AsH3 (г) | 66,4 | 223,0 | – | – | – | – | ||||
| As2O3 (т) | –656,89 | 108,32 | 35,02 | 203,34 | – | 95,65 | ||||
| AsCl3(г) | –270,34 | 328,82 | 82,09 | 1,00 | –5,94 | 75,48 | ||||
| As2O5(т) | –921,32 | 105,44 | – | – | – | 116,52 | ||||
| Au(т) | 47,40 | 23,68 | 5,19 | – | 25,36 | |||||
| AuCl3(т) | –118,4 | 164,4 | – | – | – | – | ||||
| Au2O3(т) | –13,0 | 134,0 | – | – | – | – | ||||
| Au(OH)3(т) | –277,8 | 121,0 | – | – | – | – | ||||
| B(т) | 5,86 | 16,78 | 9,04 | –7,49 | 11,09 | |||||
| BCl3(г) | –402,96 | 209,08 | 70,54 | 11,97 | –10,21 | 62,63 | ||||
| BF3(г) | –1136,58 | 254,01 | 52,05 | 28,03 | –8,87 | 50,46 | ||||
| B2O3 (т) | –1270,43 | 53,85 | 36,53 | 106,3 | –5,48 | 62,76 | ||||
| B2Н6 (г) | 38,5 | 231,8 | – | – | – | – | ||||
| Ba-a(т) | 60,67 | 22,26 | 13,81 | – | 28,28 | |||||
| BaCO3(т) | –1210,85 | 112,13 | 86,90 | 48,95 | –11,97 | 85,35 | ||||
| BaCl2(т) | –859,39 | 123,64 | 71,13 | 13,97 | – | 75,31 | ||||
| Ba(NO3)2(т) | –992,07 | 213,80 | 125,73 | 149,37 | –16,78 | 151,63 | ||||
| BaO(т) | –553,64 | 70,29 | 53,30 | 4,35 | –8,30 | 46,99 | ||||
| Ba(OH)2(т) | –943,49 | 100,83 | 70,71 | 91,63 | – | 97,91 | ||||
| BaSO4(т) | –1458,88 | 132,21 | 141,42 | 0,00 | –35,27 | 102,09 | ||||
| Be(т) | 9,54 | 19,16 | 8,87 | –4,77 | 16,44 | |||||
| BeO(т) | –598,73 | 14,14 | 35,35 | 16,74 | –13,26 | 25,56 | ||||
| BeCl2(т) | –496,0 | 75,8 | – | – | – | – | ||||
| Br2 (ж) | 152,21 | 75,69 | – | – | 75,69 | |||||
| Br2 (г) | 30,91 | 245,37 | 37,32 | 0,50 | –1,26 | 36,07 | ||||
| С(графит) | 5,74 | 16,86 | 4,77 | –8,54 | 8,54 | |||||
| С(алмаз) | 1,83 | 2,37 | 9,12 | 13,22 | –6,19 | 6,11 | ||||
| СО (г) | –110,53 | 197,55 | 28,41 | 4,10 | –0,46 | 29,14 | ||||
| СО2 (г) | –393,51 | 213,66 | 44,14 | 9,04 | –8,53 | 37,11 | ||||
| СОСl2 (г) | –219,50 | 283,64 | 67,15 | 12,03 | –9,04 | 57,76 | ||||
| CS2 (ж) | 88,70 | 151,04 | – | – | – | – | ||||
| CS2 (г) | 116,70 | 237,77 | 52,09 | 6,69 | –7,53 | 45,48 | ||||
| Ca-a(т) | 41,63 | 22,22 | 13,93 | – | 26,36 | |||||
| CaH2(т) | –188,7 | 42,0 | – | – | – | – | ||||
| CaC2-a(т) | –59,83 | 69,96 | 68,62 | 11,88 | –8,66 | 62,72 | ||||
| CaCl2(т) | –795,02 | 108,37 | 71,88 | 12,72 | –2,51 | 72,59 | ||||
| CaF2-a(т) | –1220,89 | 68,45 | 59,83 | 30,46 | 1,97 | 67,03 | ||||
| Ca(NO3)2(т) | –938,76 | 193,30 | 122,88 | 153,97 | –17,28 | 149,33 | ||||
| CaHPO4(т) | –1808,56 | 11,38 | 138,41 | 55,10 | –40,38 | 110,04 | ||||
| Ca(H2PO4)2(т) | –3114,57 | 189,54 | – | – | – | – | ||||
| Ca3(PO4)2-a(т) | –4120,82 | 235,98 | 201,84 | 166,02 | –20,92 | 227,82 | ||||
| CaS(т) | –476,98 | 56,61 | 42,68 | 15,90 | – | 47,49 | ||||
| CaSO4(т) | –1436,28 | 106,70 | 70,21 | 98,74 | – | 99,66 | ||||
| CaSO4´ ´0,5H2O(т) | –1578,9 | 130,0 | – | – | – | |||||
| CaSO4´ ´2H2O(т) | –2024,8 | 194,0 | – | – | – | |||||
| CaSiO3(т) | –1636,0 | 81,98 | – | – | – | |||||
| CaO2(т) | –651,7 | 43,1 | – | – | – | |||||
| СаСО3 (т) | –1206,83 | 91,71 | 104,52 | 21,92 | –25,94 | 83,47 | ||||
| СаО (т) | –635,10 | 38,07 | 49,63 | 4,52 | –6,95 | 42,05 | ||||
| Са(ОН)2 (т) | –985,12 | 83,40 | 105,20 | 12,01 | –19,00 | 87,49 | ||||
| Cd-a(т) | 51,76 | 22,22 | 12,30 | – | 25,94 | |||||
| CdCl2(т) | –390,79 | 115,30 | 61,25 | 40,17 | – | 73,22 | ||||
| CdO(т) | –258,99 | 54,81 | 48,24 | 6,38 | –4,90 | 43,64 | ||||
| CdS(т) | –156,90 | 71,13 | 54,00 | 3,80 | – | 47,32 | ||||
| CdSO4(т) | –934,41 | 123,05 | 77,32 | 77,40 | – | 99,62 | ||||
| Cl2 (г) | 223,00 | 37,03 | 1,05 | –2,85 | 33,93 | |||||
| Cl2O(г) | 75,73 | 266,30 | 53,18 | 3,35 | –7,78 | 45,44 | ||||
| ClO2(г) | 104,60 | 257,02 | 48,28 | 7,53 | –7,74 | 41,84 | ||||
| Cl2O7(ж) | –251,0 | – | – | – | – | |||||
| Cl2O7(г) | –285,0 | – | – | – | – | |||||
| Co-a(т) | 30,04 | 19,83 | 16,75 | – | 24,81 | |||||
| CoCl2(т) | –312,54 | 109,29 | 60,29 | 61,09 | – | 78,49 | ||||
| CoSO4(т) | –867,76 | 113,39 | 125,9 | 41,51 | – | 103,22 | ||||
| CoCO3(т) | –722,6 | – | – | – | – | – | ||||
| CoCl2(т) | –310,2 | 109,7 | – | – | – | – | ||||
| CoBr2(т) | –232,2 | 135,6 | – | – | – | – | ||||
| CoI2(т) | –102,1 | 158,2 | – | – | – | – | ||||
| Co(NO3)2(т) | –421,8 | 192,0 | – | – | – | – | ||||
| Co(NO3)2´ ´6H2О(т) | –1655,6 | – | – | – | – | – | ||||
| CoO(т) | –239,7 | 52,8 | – | – | – | – | ||||
| Co(OH)2(т) | –541,0 | 82,0 | – | – | – | – | ||||
| Co(OH)3(т) | –726,0 | 100,0 | – | – | – | – | ||||
| CoS(т) | –84,5 | – | – | – | – | – | ||||
| Co3O4(т) | –880,0 | 103,0 | – | – | – | – | ||||
| Cr (т) | 23,63 | 24,43 | 9,87 | –3,68 | 23,35 | |||||
| СrO3 (т) | –590,36 | 73,22 | 82,55 | 21,67 | –17,49 | 69,33 | ||||
| Cr2O3 (т) | –1140,56 | 81,17 | 119,36 | 9,20 | –15,65 | 104,52 | ||||
| CrCl3(т) | –556,47 | 123,01 | 79,50 | 41,21 | – | 91,80 | ||||
| CrCl2(т) | –395,7 | 115,7 | – | – | – | – | ||||
| CrO2Cl2(ж) | –598,7 | 209,2 | – | – | – | – | ||||
| Cr(OH)2(т) | –669,0 | 81,2 | – | – | – | – | ||||
| Cr(OH)3(т) | –995,0 | 95,4 | – | – | – | – | ||||
| Cr2(SO4)3(т) | –3308,0 | 287,9 | – | – | – | – | ||||
| Cr3C2(т) | –79,5 | 85,4 | – | – | – | – | ||||
| CrS(т) | –176,0 | 65,0 | – | – | – | – | ||||
| Cu (т) | 33,14 | 22,64 | 6,28 | – | 24,43 | |||||
| CuO (т) | –162,00 | 42,63 | 43,83 | 16,77 | –5,88 | 42,30 | ||||
| CuS (т) | –53,14 | 66,53 | 44,35 | 11,05 | – | 47,82 | ||||
| CuSO4 (т) | –771,10 | 109,20 | 78,53 | 71,96 | – | 98,87 | ||||
| Cu2O (т) | –173,18 | 92,93 | 56,57 | 29,29 | – | 63,64 | ||||
| Cu(NO3)2 (т) | –310,0 | 198,0 | – | – | – | – | ||||
| СuCl(т) | –134,24 | 87,02 | 38,27 | 34,38 | – | 48,53 | ||||
| CuCl2(т) | –205,90 | 108,07 | 67,44 | 17,56 | – | 71,88 | ||||
| Cu2S(т) | –79,50 | 120,92 | 39,25 | 130,54 | – | 76,32 | ||||
| CuCO3(т) | –595,4 | 88,0 | – | – | – | – | ||||
| CuI(т) | –68,0 | 96,7 | – | – | – | – | ||||
| Cu(OH)2(т) | –444,6 | 84,0 | – | – | – | – | ||||
| (CuOH)2CO3(т) | –1051,0 | 211,6 | – | – | – | – | ||||
| CuSO4´ ´5H2O(т) | –2280,8 | 300,4 | – | – | – | – | ||||
| F2(г) | 202,67 | 34,56 | 2,51 | –3,51 | 31,30 | |||||
| Fe-a(т) | 27,15 | 17,24 | 24,77 | – | 24,98 | |||||
| FeO (т) | –264,85 | 60,75 | 50,80 | 8,21 | –3,31 | 49,92 | ||||
| Fe2O3 (т) | –822,16 | 87,45 | 97,74 | 72,13 | –12,89 | 103,76 | ||||
| Fe3O4 (т) | –1117,13 | 146,19 | 86,27 | 208,92 | – | 150,79 | ||||
| FeS (т) | –100,42 | 60,29 | 50,33 | 16,31 | – | 54,81 | ||||
| FeS2 (т) | –177,40 | 52,93 | 74,81 | 5,52 | –12,76 | 62,17 | ||||
| FeCO3(т) | –738,15 | 95,40 | 48,66 | 112,13 | – | 83,26 | ||||
| FeSO4(т) | –927,59 | 107,53 | – | – | – | 100,58 | ||||
| FeSO4´ ´7H2O(т) | –3017,8 | 409,5 | – | – | – | – | ||||
| FeCl2(т) | –341,98 | 118,1 | – | – | – | – | ||||
| FeCl3(т) | –399,7 | 142,4 | – | – | – | – | ||||
| Fe(OH)2(т) | –562,1 | 88,0 | – | – | – | – | ||||
| Fe(OH)3(т) | –827,2 | 105,0 | – | – | – | – | ||||
| FeTiO3(т) | –1235,5 | 105,9 | – | – | – | – | ||||
| Fe2(SO4)3(т) | –2582,0 | 283,0 | – | – | – | – | ||||
| Fe3C(т) | 25,0 | 108,0 | – | – | – | – | ||||
| Н2 (г) | 130,52 | 27,28 | 3,26 | 0,50 | 28,83 | |||||
| HBr (г) | –36,38 | 198,58 | 26,15 | 5,86 | 1,09 | 29,14 | ||||
| HCl (г) | –92,30 | 186,70 | 26,53 | 4,60 | 1,09 | 29,14 | ||||
| НF (г) | –273,30 | 173,67 | 26,90 | 3,43 | 1,09 | 29,14 | ||||
| HI (г) | 26,36 | 206,30 | 26,32 | 5,94 | 0,92 | 29,16 | ||||
| Н2О (г) | –241,81 | 188,74 | 30,0 | 10,71 | 0,33 | 33,61 | ||||
| Н2О (ж) | –285,84 | 69,96 | 39,02 | 76,64 | 11,96 | 75,30 | ||||
| Н2О2 (ж) | –187,86 | 109,60 | 53,60 | 117,15 | – | 89,33 | ||||
| Н2S (г) | –20,60 | 205,70 | 29,37 | 15,40 | – | 33,44 | ||||
| HNO3 (ж) | –173,00 | 156,16 | – | – | – | 109,87 | ||||
| HNO3 (г) | –133,90 | 266,39 | – | – | – | 54,12 | ||||
| H2SO4(ж) | –813,99 | 156,90 | 156,90 | 28,30 | –23,46 | 138,91 | ||||
| H3PO4(ж) | –1266,90 | 200,83 | – | – | – | 106,10 | ||||
| H3BO3(т) | –1094,0 | 88,8 | – | – | – | – | ||||
| HСlO4(ж) | –34,5 | 188,4 | – | – | – | – | ||||
| H2SiO3(т) | –1189,1 | – | – | – | – | – | ||||
| H4SiO4(т) | –1480,0 | – | – | – | – | – | ||||
| HТcO4(т) | –690,5 | 139,8 | – | – | – | – | ||||
| H2WO4(т) | –1172,4 | 117,2 | – | – | – | – | ||||
| K(т) | 64,18 | 5,61 | 81,17 | 29,58 | ||||||
| KAl(SO4)2(т) | –2465,0 | 204,5 | 234,10 | 82,34 | –58,41 | 193,00 | ||||
| KBr(т) | –393,80 | 95,94 | 48,37 | 13,89 | – | 52,30 | ||||
| KI(т) | –327,90 | 106,40 | 38,84 | 28,92 | 4,93 | 53,00 | ||||
| KOH(т) | –424,72 | 79,28 | 42,66 | 76,79 | – | 65,60 | ||||
| K2CrO4(т) | –1385,74 | 200,00 | 123,72 | 74,79 | – | 146,00 | ||||
| K2Cr2O7(т) | –2067,27 | 291,21 | 153,38 | 229,29 | – | 219,70 | ||||
| K2SO4(т) | –1433,69 | 175,56 | 120,37 | 99,58 | –17,82 | 130,01 | ||||
| K2CO3(т) | –1150,18 | 155,52 | 80,29 | 109,04 | – | 114,44 | ||||
| KCl(т) | –436,68 | 82,55 | 41,38 | 21,76 | 3,22 | 51,49 | ||||
| KСlO3(т) | –391,20 | 142,97 | – | – | – | 100,25 | ||||
| KМnO4(т) | –828,89 | 171,54 | – | – | – | 117,54 | ||||
| KNO3-a(т) | –492,71 | 132,93 | 60,88 | 118,83 | – | 96,29 | ||||
| KNO2(т) | –370,6 | 152,1 | – | – | – | – | ||||
| KCl´MgCl2´ ´6H2O(т) | –2946,0 | – | – | – | – | – | ||||
| K2O2(т) | –440,0 | 120,0 | – | – | – | – | ||||
| K2S2O8(т) | –1918,0 | 278,7 | – | – | – | – | ||||
| KСlO4(т) | –427,2 | 151,0 | – | – | – | – | ||||
| K2SiO3(т) | –1590,0 | 146,1 | – | – | – | – | ||||
| K2S(т) | –387,0 | 113,0 | – | – | – | – | ||||
| KO2(т) | –283,0 | 117,0 | – | – | – | – | ||||
| K2O(т) | –363,2 | 94,1 | – | – | – | – | ||||
| K3PO4(т) | –1988,0 | 212,0 | – | – | – | – | ||||
| KF(т) | –566,0 | 66,5 | – | – | – | – | ||||
| Li(т) | 28,24 | 6,86 | 46,44 | 3,51 | 24,73 | |||||
| Li2O(т) | –595,8 | 37,87 | – | – | – | – | ||||
| Li2CO3(т) | –1215,0 | 90,2 | 42,53 | 117,34 | – | 96,20 | ||||
| LiOH(т) | –484,67 | 42,78 | 50,17 | 34,48 | 9,50 | 49,58 | ||||
| Li2SO4(т) | –1435,86 | 114,00 | 118,95 | 93,34 | –27,20 | 117,60 | ||||
| LiCl(т) | –408,27 | 59,30 | 41,42 | 23,40 | – | 48,39 | ||||
| Мg(т) | 32,68 | 22,30 | 10,64 | –0,42 | 24,89 | |||||
| MgCO3(т) | –1095,85 | 65,10 | 77,91 | 57,74 | –17,41 | 76,11 | ||||
| MgO(т) | –601,49 | 27,07 | 48,98 | 3,14 | –11,44 | 37,20 | ||||
| Mg(OH)2(т) | –924,66 | 63,14 | 46,99 | 102,85 | – | 76,99 | ||||
| MgSO4´ ´6H2O | –3089,50 | 348,10 | – | – | – | 348,10 | ||||
| MgCl2(т) | –644,80 | 89,54 | 79,08 | 5,94 | –8,62 | 71,25 | ||||
| MgSO4(т) | –1287,42 | 91,55 | 106,44 | 46,28 | –21,90 | 95,60 | ||||
| Mn(т) | 32,01 | 23,85 | 14,14 | –1,59 | 26,28 | |||||
| MnCO3(т) | –881,66 | 109,54 | 92,01 | 38,91 | –19,62 | 81,50 | ||||
| MnO(т) | –385,10 | 61,50 | 46,48 | 8,12 | –3,68 | 44,10 | ||||
| MnO2(т) | –521,49 | 53,14 | 69,45 | 10,21 | –16,23 | 54,02 | ||||
| Mn3O4(т) | –1387,60 | 154,81 | 144,93 | 45,27 | –9,20 | 148,08 | ||||
| Mn2O7(ж) | –726,00 | – | – | – | – | – | ||||
| Mn2O3(т) | –959,81 | 110,46 | 103,50 | 35,06 | –13,51 | 107,50 | ||||
| MnS(т) | –214,35 | 80,75 | 47,70 | 7,53 | – | 49,92 | ||||
| Mn(OH)2(т) | –700,00 | 95,00 | – | – | – | – | ||||
| Mn3C(т) | 5,60 | 99,00 | – | – | – | – | ||||
| MnSiO3(т) | –1321,0 | 89,00 | – | – | – | – | ||||
| MnSO4(т) | –1066,7 | 112,00 | – | – | – | – | ||||
| N2(г) | 191,50 | 27,87 | 4,27 | – | 29,12 | |||||
| NH3(г) | –45,94 | 192,66 | 29,80 | 25,48 | –1,67 | 35,16 | ||||
| NH4Cl-b(т) | –314,22 | 95,81 | 49,37 | 133,89 | – | 84,10 | ||||
| NH4NO3(т) | –365,10 | 150,60 | – | – | – | 139,33 | ||||
| NH4NO2(т) | –256,10 | – | – | – | – | – | ||||
| NO(г) | 91,26 | 210,62 | 29,58 | 3,85 | –0,59 | 29,86 | ||||
| NO2(г) | 34,19 | 240,06 | 441,16 | 11,33 | –7,02 | 36,33 | ||||
| NOCl(г) | 52,59 | 263,50 | 44,89 | 7,70 | –6,95 | 39,37 | ||||
| NF3(г) | –124,30 | 260,10 | – | – | – | – | ||||
| NOBr(г) | 82,20 | 273,50 | – | – | – | – | ||||
| NOF(г) | –66,50 | 247,90 | – | – | – | – | ||||
| NOI(г) | 100,00 | 282,80 | – | – | – | – | ||||
| N2H4(г) | 95,40 | 238,36 | – | – | – | – | ||||
| N2H4(ж) | 50,53 | 121,40 | – | – | – | – | ||||
| N2O(г) | 82,01 | 220,00 | 45,69 | 8,62 | –8,53 | 38,62 | ||||
| N2O3(г) | 83,30 | 307,30 | – | – | – | – | ||||
| N2O4(г) | 11,11 | 304,35 | 83,89 | 39,75 | –14,9 | 79,16 | ||||
| N2O5(г) | 13,30 | 355,60 | 127,45 | 16,54 | –32,85 | 95,28 | ||||
| NH4SCN (т) | –82,10 | 140,34 | – | – | – | – | ||||
| NH4ClO4(т) | –296,00 | 184,30 | – | – | – | – | ||||
| NH4HCO3(т) | –850,80 | 121,00 | – | – | – | – | ||||
| (NH4)2CO3(т) | (–929,0) | – | – | – | – | – | ||||
| (NH4)2S(т) | (–167,0) | – | – | – | – | – | ||||
| (NH4)2SO4(т) | –1180,31 | 220,08 | 103,60 | 280,80 | – | 187,30 | ||||
| (NH4)3PO4(т) | –1672,60 | (258,83) | – | – | – | – | ||||
| NH4VO3(т) | –1050,00 | 140,45 | – | – | – | – | ||||
| NH4Al(SO4)2(т) | –2347,00 | 216,20 | – | – | – | – | ||||
| (NH4)2SO4(т) | –1179,30 | 220,30 | 103,64 | 281,16 | – | – | ||||
| NH4Cr(SO4)2´´12H2O(т) | –5562,00 | 712,00 | – | – | – | – | ||||
| Ga(т) | 41,09 | 26,07 | – | – | 26,07 | |||||
| GaCl3(т) | –524,70 | 133,40 | – | – | – | – | ||||
| Ga(ОН)3(т) | –1014,60 | 84,90 | – | – | – | – | ||||
| Ge(т) | 31,09 | 25,02 | 3,43 | –2,34 | 25,35 | |||||
| GeO2(т) | –554,71 | 55,27 | 68,91 | 9,83 | –17,70 | 52,09 | ||||
| GeCl4(г) | –504,60 | 347,70 | – | – | – | – | ||||
| Hg(ж) | 75,90 | 26,94 | 0,00 | 0,79 | 27,99 | |||||
| HgCl2(т) | –228,24 | 140,02 | 69,99 | 20,28 | –1,89 | 73,91 | ||||
| Hg2Cl2(т) | –265,06 | 192,76 | 92,47 | 30,96 | – | 101,70 | ||||
| HgO(т) | –90,88 | 70,29 | 36,04 | 29,64 | – | 44,88 | ||||
| HgS(т) | –58,99 | 82,42 | 43,84 | 15,27 | – | 48,41 | ||||
| I2(т) | 116,14 | 40,12 | 49,79 | – | 54,44 | |||||
| I2(г) | 62,43 | 260,60 | 37,40 | 0,59 | –0,71 | 36,90 | ||||
| In(т) | 57,82 | 20,25 | 21,59 | – | 26,74 | |||||
| Na(т) | 51,21 | 16,82 | 37,82 | – | 28,24 | |||||
| NaAlO2(т) | –1133,03 | 70,29 | 87,95 | 17,70 | –17,74 | 73,30 | ||||
| NaCl(т) | –411,12 | 72,13 | 45,94 | 16,32 | – | 50,81 | ||||
| NaF(т) | –573,63 | 51,30 | 43,51 | 16,23 | –1,38 | 46,86 | ||||
| NaBr(т) | –361,41 | 86,82 | 47,92 | 13,31 | – | 51,90 | ||||
| NaI(т) | –287,86 | 98,32 | 48,88 | 12,05 | – | 52,50 | ||||
| NaNO3-a(т) | –466,70 | 116,50 | 25,69 | 225,94 | – | 93,05 | ||||
| NaOH-a(т) | –426,35 | 64,43 | 7,34 | 125,0 | 13,38 | 59,66 | ||||
| Na2S(т) | –374,47 | 79,50 | 82,89 | 6,86 | – | 84,93 | ||||
| Na2SO3(т) | –1089,43 | 146,02 | 107,11 | 43,51 | – | 120,08 | ||||
| Na2SO4-a(т) | –1387,21 | 149,62 | 82,32 | 154,36 | – | 128,35 | ||||
| Na2SiO3(т) | –1561,43 | 113,76 | 130,29 | 40,17 | –27,02 | 111,81 | ||||
| Na3AlF6-a(т) | –3309,54 | 283,49 | 172,27 | 158,45 | – | 219,51 | ||||
| Na3PO4(т) | –1924,64 | 224,68 | 136,10 | 67,00 | – | 153,57 | ||||
| NaHCO3(т) | –947,30 | 102,10 | 44,89 | 143,89 | – | 87,70 | ||||
| Na2CO3-a(т) | –1130,80 | 138,80 | 70,63 | 135,60 | – | 11,30 | ||||
| Na2CO3´ ´10H2O(т) | –4077,0 | 2172,00 | – | – | – | 536,0 | ||||
| Na2O(т) | –417,98 | 75,06 | 77,11 | 19,33 | –12,59 | 68,89 | ||||
| Na2O2(т) | –513,21 | 94,81 | 74,00 | 56,66 | – | 90,89 | ||||
| Ni(т) | 29,86 | 16,99 | 29,46 | – | 26,07 | |||||
| NiO-a(т) | –239,74 | 37,99 | –20,88 | 157,23 | 16,28 | 44,31 | ||||
| NiS(т) | –79,50 | 52,97 | 38,70 | 26,78 | – | 47,11 | ||||
| NiSO4´7H2O(т) | –2977,0 | 378,90 | – | – | – | – | ||||
| NiSO4(т) | –873,49 | 103,85 | 125,94 | 41,51 | – | 97,70 | ||||
| Ni(OH)2(т) | –543,00 | 79,90 | – | – | – | – | ||||
| NiCO3(т) | –694,00 | 86,20 | – | – | – | – | ||||
| Ni(NO3)2´ ´6H2O(т) | –2215,00 | 511,00 | – | – | – | – | ||||
| NiCl2(т) | –304,18 | 98,07 | 73,27 | 13,23 | – | 71,67 | ||||
| O2(г) | 205,03 | 31,46 | 3,39 | –3,77 | 29,37 | |||||
| О3(г) | 142,3 | 238,82 | 47,02 | 8,04 | –9,04 | 39,25 | ||||
| P бел.(т) | 41,09 | 23,82 | – | – | 23,82 | |||||
| P красн.(т) | –17,45 | 22,80 | 16,95 | 14,89 | – | 21,39 | ||||
| Р2(г) | 143,85 | 217,94 | 36,13 | 0,85 | –4,31 | 32,05 | ||||
| Р4О10(т) | –2984,03 | 228,86 | 93,30 | 407,19 | – | 211,71 | ||||
| Р4О6(т) | –1569,00 | – | – | – | – | – | ||||
| РН3(г) | 5,44 | 210,1 | – | – | – | – | ||||
| PCl3(г) | –287,02 | 311,71 | 80,11 | 3,10 | 7,99 | 71,84 | ||||
| PCl5(г) | –374,89 | 364,47 | 129,49 | 2,93 | –16,40 | 112,97 | ||||
| PF3(г) | –956,50 | 272,60 | – | – | – | – | ||||
| PF5(г) | –1593,00 | 293,00 | – | – | – | – | ||||
| Pb(т) | 64,81 | 24,23 | 8,71 | – | 26,82 | |||||
| PbO(т) | –217,61 | 68,70 | 37,87 | 26,78 | – | 45,77 | ||||
| PbO2(т) | –276,56 | 71,92 | 53,14 | 32,64 | – | 64,77 | ||||
| Pb3O4(т) | –723,41 | 211,29 | 177,49 | 34,39 | –29,29 | 146,86 | ||||
| PbBr2(т) | –282,42 | 161,75 | 77,78 | 9,20 | – | 80,54 | ||||
| PbCl2(т) | –359,82 | 135,98 | 66,78 | 33,47 | – | 76,99 | ||||
| PbI2(т) | –175,23 | 175,32 | 75,31 | 19,66 | – | 81,17 | ||||
| PbSO4(т) | –920,48 | 148,57 | 45,86 | 129,70 | 17,57 | 103,22 | ||||
| PbCO3(т) | –700,00 | 130,96 | 51,84 | 119,70 | – | 87,45 | ||||
| Pb(NO3)2(т) | –451,70 | 217,90 | – | – | – | – | ||||
| PbSiO3(т) | –1137,2 | 110,0 | – | – | – | – | ||||
| PbCrO4(т) | –925,0 | 167,0 | – | – | – | – | ||||
| PbS(т) | –100,42 | 91,20 | 46,74 | 9,20 | 49,48 | |||||
| Pt(т) | 41,55 | 24,02 | 5,61 | – | 25,86 | |||||
| PtCl2(т) | –106,69 | 219,79 | 67,78 | 25,98 | – | 75,52 | ||||
| PtCl4(т) | –229,28 | 267,88 | 112,21 | 129,70 | – | 150,86 | ||||
| S ромб.(т) | 31,92 | 22,68 | – | – | 22,68 | |||||
| S монокл.(т) | 0,38 | 32,55 | 23,64 | – | – | 23,64 | ||||
| S2(г) | 128,37 | 228,03 | 36,11 | 1,09 | –3,52 | 32,51 | ||||
| SO2(г) | –296,90 | 248,07 | 46,19 | 7,87 | –7,70 | 39,87 | ||||
| SOCl2(ж) | –247,00 | 278,60 | – | – | – | – | ||||
| SO2Cl2(ж) | –394,13 | 216,31 | 133,89 | – | – | 133,89 | ||||
| SO2Cl2(г) | –363,17 | 311,29 | 87,91 | 16,15 | –14,23 | 77,40 | ||||
| SO3(г) | –395,85 | 256,69 | 64,98 | 11,75 | –16,37 | 50,09 | ||||
| Sb(т) | 45,69 | 23,10 | 7,28 | – | 25,23 | |||||
| Sb2O3(т) | –715,46 | 132,63 | 92,05 | 66,11 | – | 111,76 | ||||
| Sb2O5(т) | –1007,51 | 125,10 | 45,86 | 241,04 | – | 117,61 | ||||
| Sb2S3(т) | –157,74 | 181,59 | 101,29 | 55,23 | – | 123,22 | ||||
| Si(т) | 18,83 | 22,82 | 3,86 | –3,54 | 19,99 | |||||
| SiO2 кварц-a(т) | –910,94 | 41,84 | 46,99 | 34,31 | –11,3 | 44,43 | ||||
| SiCl4(ж) | –687,85 | 239,74 | 145,27 | – | – | 145,27 | ||||
| SiF4(г) | –1614,94 | 282,38 | 91,46 | 13,26 | –19,66 | 73,64 | ||||
| SiH4(г) | 33,73 | 204,56 | 46,26 | 36,76 | –12,77 | 42,89 | ||||
| Si2H6(г) | 71,50 | 274,50 | – | – | – | – | ||||
| Sn(т) | 51,55 | 21,59 | 18,10 | – | 26,99 | |||||
| SnS(т) | –110,17 | 77,00 | 35,69 | 31,30 | 3,77 | 49,25 | ||||
| SnO(т) | –286,00 | 56,48 | 39,96 | 14,64 | – | 44,35 | ||||
| SnO2(т) | –580,74 | 52,34 | 73,89 | 10,04 | –21,59 | 52,59 | ||||
| SnCl2(т) | –330,95 | 131,80 | 50,63 | 83,68 | – | 75,58 | ||||
| SnCl4(ж) | –528,86 | 258,99 | 165,27 | – | – | 165,27 | ||||
| SnH4(г) | 16,30 | 228,70 | – | – | – | – | ||||
| Sn(OH)2(т) | –506,30 | 87,70 | – | – | – | – | ||||
| Sr(т) | 55,69 | 22,22 | 13,89 | – | 26,36 | |||||
| SrO(т) | –592,04 | 54,39 | 50,75 | 5,27 | –6,49 | 45,03 | ||||
| SrSO4(т) | –1444,74 | 117,57 | 91,20 | 55,65 | – | 107,79 | ||||
| Те(т) | 49,50 | 19,12 | 22,09 | – | 25,71 | |||||
| ТеО2(т) | –323,42 | 74,06 | 65,19 | 14,56 | –5,02 | 63,88 | ||||
| Ti(т) | 30,63 | 21,10 | 10,54 | – | 25,02 | |||||
| TiCl4(г) | –763,16 | 354,80 | 107,18 | 0,47 | –10,55 | 95,45 | ||||
| TiO2 рутил(т) | –944,75 | 50,33 | 62,86 | 11,36 | –9,96 | 55,04 | ||||
| Ti2O3(т) | –1520,50 | 78,80 | – | – | – | – | ||||
| Tl-a(т) | 64,18 | 22,01 | 14,48 | – | 26,32 | |||||
| Tl2O(т) | –167,36 | 134,31 | 56,07 | 41,84 | – | 68,54 | ||||
| Tl2O3(т) | –390,40 | 148,10 | – | – | – | – | ||||
| TlCl(т) | –204,18 | 111,29 | 50,21 | 8,37 | – | 52,70 | ||||
| V(т) | 28,90 | – | – | – | – | |||||
| VCl3(т) | –582,40 | 131,00 | – | – | – | – | ||||
| VO(т) | –418,00 | 38,90 | – | – | – | – | ||||
| V2O3(т) | –1238,00 | 98,30 | – | – | – | – | ||||
| V2O5(т) | –1573,00 | 130,96 | – | – | – | – | ||||
| W(т) | 32,64 | 22,91 | 4,69 | – | 24,27 | |||||
| WO3(т) | –843,00 | 75,90 | 87,65 | 16,17 | –17,50 | 72,79 | ||||
| Zn(т) | 41,63 | 22,38 | 10,04 | – | 25,44 | |||||
| ZnO(т) | –348,11 | 43,51 | 48,99 | 5,10 | –9,12 | 40,25 | ||||
| ZnS(т) | –205,18 | 57,66 | 49,25 | 5,27 | –4,85 | 45,36 | ||||
| ZnCO3(т) | –812,53 | 80,33 | 38,91 | 138,07 | – | 80,08 | ||||
| ZnSO4(т) | –981,36 | 110,54 | 76,36 | 76,15 | – | 99,06 | ||||
| Zr-a(т) | 38,99 | 21,97 | 11,63 | – | 25,44 | |||||
| ZrCl4(т) | –979,77 | 181,42 | 124,97 | 14,14 | –8,37 | 119,77 | ||||
| ZrO2-a(т) | –1097,46 | 50,36 | 69,62 | 7,53 | –14,06 | 56,05 | ||||
| Органические соединения | ||||||||||
| Формула для вычисления теплоемкости Ср 0 = а + bТ + сТ 2 | ||||||||||
| метан СН4 (г) | –74,85 | 186,27 | 14,32 | 74,66 | –17,43 | 35,71 | ||||
| ацетилен С2Н2 (г) | 226,75 | 200,82 | 26,44 | 66,65 | –26,48 | 43,93 | ||||
| этилен С2Н4 (г) | 52,30 | 219,45 | 11,32 | 122,01 | –37,90 | 43,56 | ||||
| этан С2Н6(г) | –84,67 | 229,49 | 5,75 | 175,11 | –57,85 | 52,64 | ||||
| пропан С3Н8 (г) | –103,85 | 269,91 | 1,72 | 270,75 | –94,48 | 73,51 | ||||
| бензол С6Н6 (ж) | 49,04 | 173,26 | 59,50 | 255,02 | – | 135,14 | ||||
| бензол С6Н6 (г) | 82,93 | 269,20 | –21,09 | 400,12 | –169,87 | 81,67 | ||||
| циклогексан С6Н12 (г) | –123,14 | 298,24 | –51,72 | 598,77 | –230,0 | 106,27 | ||||
| толуол С7Н8 (г) | 50,00 | 320,66 | –21,59 | 476,85 | –190,3 | 103,64 | ||||
| муравьиный альдегид СН2О(г) | –115,90 | 218,78 | 18,82 | 58,38 | –15,61 | 35,39 | ||||
| муравьиная кислота СН2О2 (г) | –378,80 | 248,77 | 19,40 | 112,80 | –47,50 | 45,80 | ||||
| щавелевая кислота Н2С2О4 (т) | –829,94 | 120,08 | – | – | – | 109,00 | ||||
| оксид этилена С2Н4О (г) | –52,63 | 242,42 | –2,02 | 190,60 | –73,60 | 48,50 | ||||
| этиловый спирт С2Н5ОН (ж) | –276,98 | 160,67 | – | – | – | 11,96 | ||||
| этиловый спирт С2Н5ОН (г) | –234,80 | 281,38 | 10,99 | 204,70 | –74,20 | 65,75 | ||||
| этиленгликоль С2Н6О2 (ж) | –454,90 | 167,32 | – | – | – | 151,00 | ||||
| этиленгликоль С2Н6О2 (г) | –389,32 | 323,55 | 44,26 | 200,50 | –77,90 | 93,30 | ||||
метиловый спирт  СН3ОН (ж) СН3ОН (ж)
| –238,57 | 126,78 | – | – | – | 81,60 | ||||
| метиловый спирт СН3ОН (г) | –201,00 | 236,76 | 15,28 | 105,20 | –31,04 | 44,13 | ||||
| пропанол–1 C3H8O (г) | –257,53 | 324,80 | 13,10 | 277,50 | –98,44 | 87,11 | ||||
| уксусная кислота СН3СООН(ж) | –484,09 | 159,83 | – | – | – | 123,46 | ||||
| уксусная кислота СН3СООН (г) | –434,84 | 282,50 | 14,82 | 196,70 | –77,70 | 66,50 | ||||
| ацетон (СН3)2СО (ж) | –248,11 | 200,41 | – | – | – | 125,00 | ||||
| ацетон (СН3)2СО (г) | –217,57 | 294,93 | 22,47 | 201,80 | –63,50 | 74,90 | ||||
| глицерин С3Н8О3 (ж) | –668,60 | 204,47 | – | – | – | 223,01 | ||||
| фенол С6Н5ОН (т) | –164,85 | 144,01 | – | – | – | 134,70 | ||||
| диметиловый эфир (СН3)2О (г) | –184,05 | 267,06 | 16,18 | 183,90 | –58,70 | 65,81 | ||||
| сахароза С12Н22О11(т) | –2222,12 | 360,24 | – | – | – | 425,00 | ||||
| тетрахлорметан ССl4 (г) | –100,42 | 310,12 | 59,36 | 97,00 | –49,57 | 83,76 | ||||
| тетрахлорметан ССl4 (ж) | –132,84 | 216,19 | – | – | – | 131,70 | ||||
| хлороформ СНСl3 (ж) | –132,21 | 202,92 | – | – | – | 116,30 | ||||
| хлорофлорм CHCl3 (г) | –101,25 | 295,64 | 29,50 | 148,90 | –90,70 | 65,73 | ||||
| пиридин C5H5N (ж) | 99,95 | 177,90 | – | – | – | 132,72 | ||||
| пиридин C5H5N (г) | 140,16 | 282,80 | –18,45 | 370,10 | –154,3 | 78,12 | ||||
| анилин C6H5NH2 (ж) | 31,09 | 191,29 | – | – | – | 190,79 | ||||
| анилин C6H5NH2 (г) | 86,86 | 319,20 | –6,00 | 439,40 | –185,3 | 108,40 | ||||
| мочевина (NH2)2CO (т) | –333,17 | 104,60 | – | – | – | 93,14 | ||||
нитробензол
 C6H5NO2 (ж) C6H5NO2 (ж)
| 15,90 | 224,26 | – | – | – | 186,00 | ||||
Не нашли, что искали? Воспользуйтесь поиском: