
ТОР 5 статей:
Методические подходы к анализу финансового состояния предприятия
Проблема периодизации русской литературы ХХ века. Краткая характеристика второй половины ХХ века
Характеристика шлифовальных кругов и ее маркировка
Служебные части речи. Предлог. Союз. Частицы
КАТЕГОРИИ:
- Археология
- Архитектура
- Астрономия
- Аудит
- Биология
- Ботаника
- Бухгалтерский учёт
- Войное дело
- Генетика
- География
- Геология
- Дизайн
- Искусство
- История
- Кино
- Кулинария
- Культура
- Литература
- Математика
- Медицина
- Металлургия
- Мифология
- Музыка
- Психология
- Религия
- Спорт
- Строительство
- Техника
- Транспорт
- Туризм
- Усадьба
- Физика
- Фотография
- Химия
- Экология
- Электричество
- Электроника
- Энергетика
Задачи для самостоятельного решения 4 страница
Приложение 13
Термодинамические константы некоторых ионов
в водных растворах
| Ион |  кДж/моль
кДж/моль
|  кДж/моль
кДж/моль
| 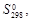 Дж/(моль×К)
Дж/(моль×К)
|
| Ag+ | 105,478 | 77,05 | 72,732 |
| [Ag(NH3)2]+ | – | –17,389 | 241,6 |
| [Ag(OH)2]– | – | –260,748 | – |
| [Ag(S2O3)2]3– | – | –1034,968 | – |
| Al3+ | –524,7 | –481,2 | –313,4 |
| [AlF6]3– | – | 2255,53 | –167,2 |
| [Al(OH)4]– | – | –1270,26 | – |
| AlO2– | – | –855,65 | – |
| H2AlO3– | – | –1066,74 | – |
| AsO2– | –426,36 | –349,699 | 41,382 |
| AsO43– | –887,289 | –647,9 | –162,6 |
| HasO42– | –905,47 | –714,03 | 3,762 |
| H2AsO3– | –714,111 | –586,663 | 110,35 |
| BO2– | – | –708,928 | –83,6 |
| BeO22– | – | –649,154 | –112,86 |
| Bi3+ | – | 82,764 | – |
| BiOH2+ | – | –146,3 | – |
| Ba2+ | –538,36 | –560,7 | |
| Br– | –120,92 | –102,82 | 80,71 |
| BrO3– | –83,60 | 45,6 | 161,1 |
| CH3COO– | –488,87 | –375,39 | – |
| CN– | 151,0 | 165,7 | 92,0 |
| CNS– | 76,37 | 92,587 | 144,21 |
| CO32– | –676,26 | –528,10 | –53,1 |
| C2O42– | –824,2 | –674,9 | 51,0 |
| Ca2+ | –542,96 | –553,04 | –55,2 |
| Cd2+ | –72,38 | –77,74 | –61,1 |
| CdOH+ | – | –269,53 | – |
| Cl– | –167,46 | –131,17 | 55,10 |
| ClO– | –107,65 | –38,53 | 47,53 |
| ClO2– | –69,0 | 14,6 | 100,4 |
| ClO3– | –98,32 | –2,59 | 163,2 |
| ClO4– | –131,42 | –10,75 | 180,7 |
| Co2+ | –67,4 | –51,5 | –111,7 |
| Co3+ | – | 120,8 | –111,61 |
| [Co(NH3)6]2+ | – | –232,83 | – |
| [Co(NH3)6]3+ | – | –230,74 | – |
| Cr2+ | – | –175,98 | – |
| Cr3+ | –231,99 | – | – |
| CrO2– | – | –522,5 | – |
| CrO42– | –863,2 | –706,3 | 38,5 |
| Cr2O72– | –1479,72 | –1318,37 | – |
| HСrO4– | –920,44 | –772,88 | – |
| С+ | –247,7 | –282,04 | 133,1 |
| Cu+ | 71,5 | 50,2 | 39,3 |
| Cu2+ | 64,39 | 64,98 | –98,7 |
| [Cu(NH3)]+ | – | –11,7 | – |
| [Cu(NH3)2]+ | – | – | 26,33 |
| F– | –329,11 | –276,48 | –9,6 |
| Fe2+ | –87,9 | –84,94 | –113,4 |
| Fe3+ | –47,7 | –10,54 | –293,3 |
| [Fe(CN)6]3– | 634,52 | 802,98 | 267,52 |
| H+ | |||
| H3O+ | –285,53 | – | – |
| HCOO– | –410,0 | –334,7 | 91,6 |
| HCO3– | –691,11 | –587,06 | 95,0 |
| Hg22+ | 174,01 | 164,77 | –22,0 |
| Hg2+ | 168,3 | 154,18 | 74,1 |
| [Hg(CN)4]2– | – | 590,63 | – |
| HgCl42– | – | –450,19 | – |
| HgI42– | – | –172,22 | – |
| IO– | – | –34,69 | – |
| IO3– | –230,1 | –135,6 | 115,9 |
| IO4– | –147,49 | – | – |
| I– | –55,94 | –51,67 | 109,37 |
| K+ | –251,21 | –282,28 | 102,5 |
| Li+ | –278,46 | –293,80 | 14,2 |
| Mg2+ | –461,96 | –456,01 | –118,0 |
| Mn2+ | –218,8 | –223,4 | –79,9 |
| Mn3+ | –112,86 | –81,93 | – |
| MnO4– | –518,4 | –425,1 | 190,0 |
| MnO42– | – | –503,27 | – |
| HMnO2– | – | –505,36 | – |
| Mo2+ | – | –57,68 | – |
| MoO42– | –995,68 | –843,52 | – |
| NH4+ | –132,80 | –79,50 | 112,84 |
| NO2– | –106,3 | –35,35 | 125,1 |
| NO3– | –206,57 | –110,50 | –146,4 |
| Na+ | –239,66 | –261,87 | 60,2 |
| Ni2+ | –64,0 | –64,4 | –123,0 |
| O2– | –50,16 | – | 143,21 |
| OH– | –220,94 | –157,30 | –10,54 |
| HPO42– | –1298,7 | –1094,1 | –36,0 |
| H2PO4– | –1302,5 | –1135,1 | 89,1 |
| PO43– | –1284,1 | –1025,5 | –218 |
| H2PO3– | –968,51 | –845,41 | 79,42 |
| Pb2+ | 1,63 | –24,31 | 21,3 |
| [PtCl4]2– | –511,63 | – | – |
| [Pt(NH3)3Cl]+ | –410,06 | – | – |
| Rb+ | –246,4 | –282,21 | 124,3 |
| ReO4– | –786,68 | –698,48 | 201,89 |
| S2– | 41,8 | 83,7 | –26,8 |
| SO32– | –643,94 | –485,3 | –29,26 |
| SO42– | –907,51 | –742,99 | 17,2 |
| S2O32– | –643,72 | –512,89 | 62,7 |
| S2O42– | –745,29 | –599,41 | 117,04 |
| S2O52– | –972,27 | –790,02 | 104,5 |
| S2O72– | –1399,88 | – | – |
| S2O82– | –1337,6 | –1109,37 | 247,87 |
| S4O62– | –1205,51 | –1021,17 | 150,48 |
| HS– | –17,66 | 12,59 | 61,1 |
| HSO3– | –627,98 | –527,31 | 132,38 |
| HSO4– | –885,75 | –752,87 | 126,86 |
| Se2– | 73,99 | 129,16 | 173,47 |
| SeO32– | –508,71 | –369,51 | 12,54 |
| SeO42– | –598,58 | –691,79 | 22,91 |
| HSe– | 15,88 | 43,89 | 79,42 |
| HSeO3– | –514,06 | –411,14 | 138,36 |
| HSeO4– | –581,02 | –451,86 | 149,23 |
| Sr2+ | –545,51 | –557,3 | –26,4 |
| Te2- | – | 220,29 | – |
| TeO32– | –532,11 | –451,44 | – |
| TeO42– | –716,87 | – | – |
| HTe– | – | 157,59 | – |
| Ti2+ | – | –313,92 | – |
| Ti3+ | – | –349,45 | – |
| TiO2+ | – | –576,84 | – |
| V2+ | – | –228,65 | – |
| V3+ | – | –253,31 | – |
| VO2+ | – | –455,6 | – |
| VO22+ | – | –455,6 | –56,01 |
| WO42- | –1114,39 | –919,6 | 62,7 |
| Zn2+ | –152,42 | –147,21 | –106,48 |
| [Zn(NH3)4]2+ | –67,63 | –53,59 | – |
| ZnOH+ | – | –329,38 | – |
| [Zn(OH)4]2- | – | –862,13 | – |
Приложение 14
Энергии разрыва связей при температуре 25 °С
| Связь | Энергия связи, кДж/моль | Связь | Энергия связи, кДж/моль | Связь | Энергия связи, кДж/моль | ||
| H–H | N–O | O–Br | |||||
| O=O | N=O | Si–Si | |||||
| NºN | C–C | P–P | |||||
| C=O оксид углерода (II) | C=C | Se–Se | |||||
| F–F | CºC | H–Si | |||||
| Cl–Cl | C–N | Si–C | |||||
| Br–Br | C=N | Si–O | |||||
| I–I | CºN | S=O | |||||
| H–F | C–O | Si–F | |||||
| H–Cl | C=O оксид углерода (IV) | Si–Cl | |||||
| H–Br | C=O формальдегид | Si–Br | |||||
| H–I | C–F | Cl–F | |||||
| C–H | C–Cl | P–F | |||||
| N–H | C–Br | Br–F | |||||
| O–H | C–I | P–Cl | |||||
| S–H | C–S | PºP | |||||
| P–H | C=S | S=S | |||||
| N–N | N–F | Cграфит–C | |||||
| N=N | N–Cl | B–B | |||||
| O–O | O–F | B–F | |||||
| S–S | O–Cl |
Приложение 15
Функции  для некоторых веществ, кДж/моль
для некоторых веществ, кДж/моль
| Вещество | Температура, К | |||||||
| H2O | 0,084 | 3,743 | 6,945 | 10,544 | 14,226 | 18,033 | 21,966 | 26,024 |
| H2 | 0,054 | 2,958 | 5,883 | 8,812 | 11,749 | 14,703 | 17,673 | 20,677 |
| O2 | 0,054 | 3,025 | 6,084 | 9,242 | 12,502 | 15,841 | 19,246 | 22,707 |
| CO2 | 0,084 | 4,017 | 8,326 | 12,929 | 17,782 | 22,803 | 28,033 | 33,388 |
| CO | 0,084 | 3,012 | 5,941 | 8,953 | 12,050 | 15,188 | 18,410 | 21,714 |
| COCl2 | 0,126 | 6,234 | 12,845 | 19,790 | 27,029 | 34,434 | 42,007 | 49,706 |
| HCl | 0,084 | 2,971 | 8,889 | 8,870 | 11,841 | 14,853 | 17,949 | 21,046 |
| NH3 | 0,084 | 3,807 | 7,824 | 12,217 | 16,862 | 21,840 | 27,112 | 32,635 |
| NH4Cl | 0,159 | 9,680 | 23,470 | 33,090 | 43,082 | – | – | – |
| Na2CO3 | 0,205 | 11,996 | 26,295 | 40,593 | 57,612 | 74,630 | 91,251 | 107,872 |
| NaOH | 0,109 | 6,350 | 13,300 | 34,200 | 42,748 | 51,296 | 59,727 | 68,157 |
| S2 | 0,042 | 4,640 | 8,565 | 12,150 | 15,498 | 73,341 | 75,174 | 77,019 |
| H2S | 0,084 | 3,556 | 7,196 | 11,046 | 15,021 | 19,205 | 23,514 | 28,033 |
Приложение 16
Температуры возгонки (кипения) некоторых
неорганических веществ при давлении 101,3 кПа
| Вещество | T возг.(кип.),°С | T плавл., °С | Вещество | T возг.(кип.),°С | T плавл., °С | |
| AlCl3 | 108,2 () | 192,4 | N2H4 | 113,6 | 0,7 | |
| AsCl3 | 130,4 | –18,0 | Na | 892,0 | 97,5 | |
| BCl3 | 12,7 | –107,0 | P | 280,0 | 44,1 | |
| BeB2H8 | 90,0 (т) | 123,0 | PCl3 | 74,2 | –11,8 | |
| BeCl2 | 487,0 | 405,0 | PCl5 | 162,0 (т) | 167,0 | |
| Br2 | 58,2 | –7,3 | SO2 | –10,0 | –73,2 | |
| Cl2 | –33,8 | –100,7 | SO2Cl2 | 69,2 | –54,1 | |
| HgCl2 | 304,0 | 277,0 | SO3 | 51,6 (т) | 62,1 | |
| HNO3 | 83,8 | –41,7 | SiCl4 | 56,8 | –68,8 | |
| H2O | 100,0 | 0,0 | TiCl4 | 136,0 | –30,0 | |
| H2O2 | 158,0 (разл.) | –0,9 | He | –268,8 | – | |
| H2SO4 | 330,0 | 10,5 | Ne | –245,9 | –248,5 | |
| I2 | 183,0 | 112,9 | Ar | –185,7 | –189,2 | |
| NH2OH | 110,0 | 34,0 | Kr | –152,2 | –156,4 | |
| NH3 | –33,6 | –77,7 | Xe | –106,9 | –111,7 |
Приложение 17
Температуры плавления и кипения некоторых
органических веществ при давлении 101,3 к Па
| Вещество | T кип.,°С | T плавл., °С | |
| Формула | Название | ||
| CCl2F2 | Дихлордифторметан | –29,8 | –160 |
| CCl2O | Фосген | 8,3 | –104 |
| CCl3F | Трихлорфторметан | 23,7 | –111 |
| CCl4 | Тетрахлорметан | 76,8 | –22,9 |
| CHClF2 | Хлордифторметан | –40,8 | –160 |
| CHCl2F | Дихлорфторметан | 8,9 | –135 |
| CHCl3 | Трихлорметан | 61,3 | –63,5 |
| CHN | Цианистый водород | 25,8 | –14 |
| CH2Cl2 | Дихлорметан | 40,7 | –96,7 |
| CH2O2 | Муравьиная кислота | 100,7 | 8,2 |
| CH4O | Метанол | 64,5 | –97,9 |
| CO2 | Диоксид углерода | –78,2 (т) | –57,5 |
| CS2 | Сероуглерод | 46,2 | –111,9 |
| C2Cl4 | Тетрахлорэтилен | 120,8 | –19,0 |
| C2HCl3O2 | Трихлоуксусная кислота | 195,6 | |
| C2H2Cl2O2 | Дихлоруксусная кислота | 194,4 | 9,7 |
| C2H3ClO2 | Хлоруксусная кислота | 189,5 | 61,2 |
| C2H3N | Ацетонитрил | 81,6 | –44,9 |
| C2H4O2 | Уксусная кислота | 118,1 | 16,8 |
| C2H4Cl2 | 1,2–Дихлорэтан | 83,5 | –35,9 |
| C2H6O | Этанол | 78,4 | –114,5 |
| C2H6O2 | Этиленгликоль | 197,3 | –15,6 |
| C3H6O | Ацетон | 56,2 | –95,4 |
| C3H6O2 | Метилацетат | 57,8 | –98,7 |
| C3H6O2 | Пропионовая кислота | 141,1 | –22 |
| C3H8O | Пропанол | 97,2 | –126,2 |
| C3H8O3 | Глицерин | 290,0 | 17,9 |
| C4H8O2 | Масляная кислота | 163,5 | –4,7 |
| C4H8O2 | 1,4–Диоксан | 101,3 | 11,8 |
| C4H8O2 | Этилацетат | 77,2 | –83,6 |
| C4H10O | Диэтиловый эфир | 34,6 | –116,3 |
| C4H10O | Бутанол | 117,5 | –79,9 |
| C4H12Pb | Тетраметилсвинец | 110,0 | –27,5 |
| C5H5N | Пиридин | 115,3 | –41,8 |
| C5H12O | Амиловый спирт | 137,8 | –78,8 |
| C6H5Br | Бромбензол | 156,2 | –30,7 |
| C6H5Cl | Хлорбензол | 132,2 | –45,2 |
| C6H5I | Иодбензол | 188,6 | –28,5 |
| C6H5NO2 | Нитробензол | 210,9 | 5,8 |
| C6H6 | Бензол | 80,1 | 5,5 |
| C6H6O | Фенол | 181,9 | 40,6 |
| C6H7N | Анилин | 184,4 | –6,2 |
| C6H12 | Циклогексан | 80,7 | 6,6 |
| C6H14 | Гексан | 68,7 | –95,3 |
| C7H6O2 | Бензойная кислота | 249,2 | 121,7 |
| C7H8 | Толуол | 110,6 | –95,0 |
| C7H8O | Бензиловый спирт | 204,7 | –15,3 |
| C7H16 | Гептан | 98,4 | –90,6 |
| C8H8O | Ацетофенон | 202,4 | 20,5 |
| C8H10 | Этилбензол | 136,2 | –94,9 |
| C8H18 | Октан | 125,6 | –56,8 |
| C8H20Pb | Тетраэтилсвинец | 183,0 | –136,0 |
| C10H8 | Нафталин | 217,9 | 80,2 |
| C10H16O | Камфора | 209,2 | 178,5 |
| C12H10 | Дифенил | 254,9 | 69,5 |
| C14H10 | Антрацен | 342,0 | 217,5 |
Приложение 18
Периодическая система элементов




Учебное издание
Бахирева О.И., Соколова М.М.,
Л.С. Пан, Н.Б. Ходяшев
Не нашли, что искали? Воспользуйтесь поиском: